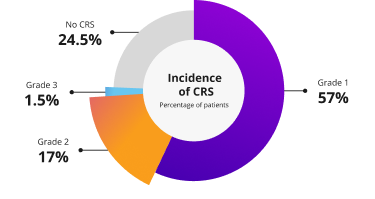
In the clinical trial primary analysis, CRS occurred in 76% of patients (N=339) who received TALVEY® at the recommended dosages
CRS was primarily Grade 1 or 2, with Grade 3 events occurring in 1.5% of patients
- Median time to onset: 27 hours (range: 0.1–167) from the last dose
- Median duration: 17 hours (range: 0–622)
- Recurrent CRS occurred in 30% of patients

Most instances of CRS occurred during step-up dosing1
| CRS experienced after each dose of TALVEY® (N=339) | |
|---|---|
| Q2W | QW |
| Step-up dosing schedule | |
| Step-up dose 1: 29% | |
| Step-up dose 2: 44% | |
| Step-up dose 3 (n=153): 33% | First treatment dose: 30% |
| First treatment dose: 12% | N/A |
| Dosing after step-up schedule | |
| Each remaining dose in Cycle 1: <3% | |
| Cumulatively from Cycle 2 onwards: <3% | |
You are now viewing a post hoc analysis of patients with RRMM treated with TALVEY® in MonumenTAL-1. This information is not included in the current full Prescribing Information and should be interpreted with caution.
The following data are from a publication characterizing the incidence and management of CRS in the MonumenTAL-1 study (N=339)2
The median time to onset and median duration of CRS events were similar across cohorts. The median follow-up time was 12.7 months for T-cell redirection–naïve Q2W patients, 18.8 months for T-cell redirection–naïve QW patients, and 14.8 months for T-cell redirection–exposed patients.
| Median time to onset | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| T-cell redirection–naïve Q2W | T-cell redirection–naïve QW | T-cell redirection–exposed | |||||||||
| 28.0 hours (range: 0.1–333.4) | 25.9 hours (range: 1.3–165.0) | 26.3 hours (range: 4.9–97.2) | |||||||||
| Median duration | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| T-cell redirection–naïve Q2W | T-cell redirection–naïve QW | T-cell redirection–exposed | |||||||||
| 18.0 hours (range: 0.0–621.8) | 14.5 hours (range: 0.5–221.6) | 20.4 hours (range: 0.9–71.5) | |||||||||
Median time to onset
T-cell redirection–naïve Q2W
28.0 hours
(range: 0.1–333.4)
T-cell redirection–naïve QW
25.9 hours
(range: 1.3–165.0)
T-cell redirection–exposed
26.3 hours
(range: 4.9–97.2)
Median duration
T-cell redirection–naïve Q2W
18.0 hours
(range: 0.0–621.8)
T-cell redirection–naïve QW
14.5 hours
(range: 0.5–221.6)
T-cell redirection–exposed
20.4 hours
(range: 0.9–71.5)
During the first 8 days of treatment, incidences of Grade 2 or 3 CRS were ≤12%
- Most patients experienced CRS events following the first 2 step-up doses
- Recurrent CRS occurred in 30% of patients
Incidence of CRS by grade and timing2
| Incidence of CRS by grade and timing2 | |||
|---|---|---|---|
| Patients with CRS events, n (%) | T-cell redirection– naïve Q2W (n=145) | T-cell redirection– naïve QW (n=143) | T-cell redirection– exposed QW (n=51) |
| Total patients with CRS | 108 (74.5) | 113 (79.0) | 39 (76.5) |
| Grade 1 CRS | |||
| Step-up dose 1 | 33 (22.8) | 41 (28.7) | 9 (17.6) |
| Step-up dose 2 | 48 (33.1) | 59 (41.3) | 15 (29.4) |
| Step-up dose 3 | 45 (31.0) | N/A | 0 |
| Cycle 1 Day 1 | 16 (11.0) | 34 (23.8) | 13 (25.5) |
| Cycle 1 Day 8 | N/A | 5 (3.5) | 1 (2.0) |
| Cycle 1 Day 15 | 7 (4.8) | 2 (1.4) | 0 |
| Cycle 1 Day 22 | N/A | 6 (4.2) | 0 |
| Cycle ≥2 | 5 (3.4) | 5 (3.5) | 2 (3.9) |
| Grade 2 CRS | |||
| Step-up dose 1 | 7 (4.8) | 7 (4.9) | 4 (7.8) |
| Step-up dose 2 | 12 (8.3) | 12 (8.4) | 6 (11.8) |
| Step-up dose 3 | 4 (2.8) | N/A | 1 (2.0) |
| Cycle 1 Day 1 | 3 (2.1) | 5 (3.5) | 3 (5.9) |
| Cycle 1 Day 8 | N/A | 0 | 0 |
| Cycle 1 Day 15 | 0 | 0 | 0 |
| Cycle 1 Day 22 | N/A | 0 | 0 |
| Cycle ≥2 | 0 | 0 | 0 |
| Grade 3 CRS | |||
| Step-up dose 1 | 0 | 0 | 0 |
| Step-up dose 2 | 0 | 1 (0.7) | 0 |
| Step-up dose 3 | 1 (0.7) | N/A | 0 |
| Cycle 1 Day 1 | 0 | 1 (0.7) | 1 (2.0) |
| Cycle 1 Day 8 | N/A | 1 (0.7) | 0 |
| Cycle 1 Day 15 | 0 | 0 | 0 |
| Cycle 1 Day 22 | N/A | 0 | 0 |
| Cycle ≥2 | 0 | 0 | 0 |
| CRS leading to discontinuation | 1 (0.7) | 0 | 0 |
CRS, cytokine release syndrome; QW, once weekly; Q2W, every 2 weeks.

