TALVEY® can cause serious, life-threatening, or fatal infections. Monitor for signs and symptoms of infection; treat appropriately. Withhold or consider permanent discontinuation based on severity.
You are now viewing a post hoc analysis of patients with RRMM treated with TALVEY® in MonumenTAL-1. This information is not included in the current full Prescribing Information and should be interpreted with caution.
Study design of the MonumenTAL-1 post hoc analysis
Study design:
This was a post hoc analysis of MonumenTAL-1 based on a data cutoff date of January 17, 2023.
Assessments:
Descriptive statistics were generated for infection type, incidence, severity, timing, and management. Patients underwent frequent monitoring for the presence of infections, which were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03. CD19+ mature B cells were assessed from whole blood using flow cytometry.
Blood samples were used for hematology laboratory tests, including hemoglobin, white blood cell count with differential, platelet count, absolute neutrophil count, and absolute lymphocyte count. GPRC5D protein expression in bone marrow samples was assessed by flow cytometry in a separate analysis of healthy donors and patients with MM.
Results:
339 patients treated with TALVEY® were included in this analysis; 288 patients (143 receiving 0.4 mg/kg QW and 145 receiving 0.8 mg/kg Q2W) had no prior T-cell redirection therapy exposure, and 51 patients (receiving either RP2D) had prior T-cell redirection therapy exposure. Median follow-up was 18.8, 12.7, and 14.8 months in the QW, Q2W, and prior T-cell redirection therapy cohorts, respectively. Baseline characteristics were similar across the 3 cohorts.
Limitations:
Limitations of MonumenTAL-1 included that it was a single-arm study: the lack of a placebo arm makes it difficult to distinguish between infection risk due to the disease course or due to TALVEY® treatment, while the lack of comparator arms prohibits comparisons of infection risk between treatment regimens. In addition, based on differences in practices across institutions involved in MonumenTAL-1, correlation with outcomes in centers utilizing varying institutional standards for infection prevention and management may be challenging.
Incidence of infections in patients treated with TALVEY®
Incidence of Grade 3 infection over time
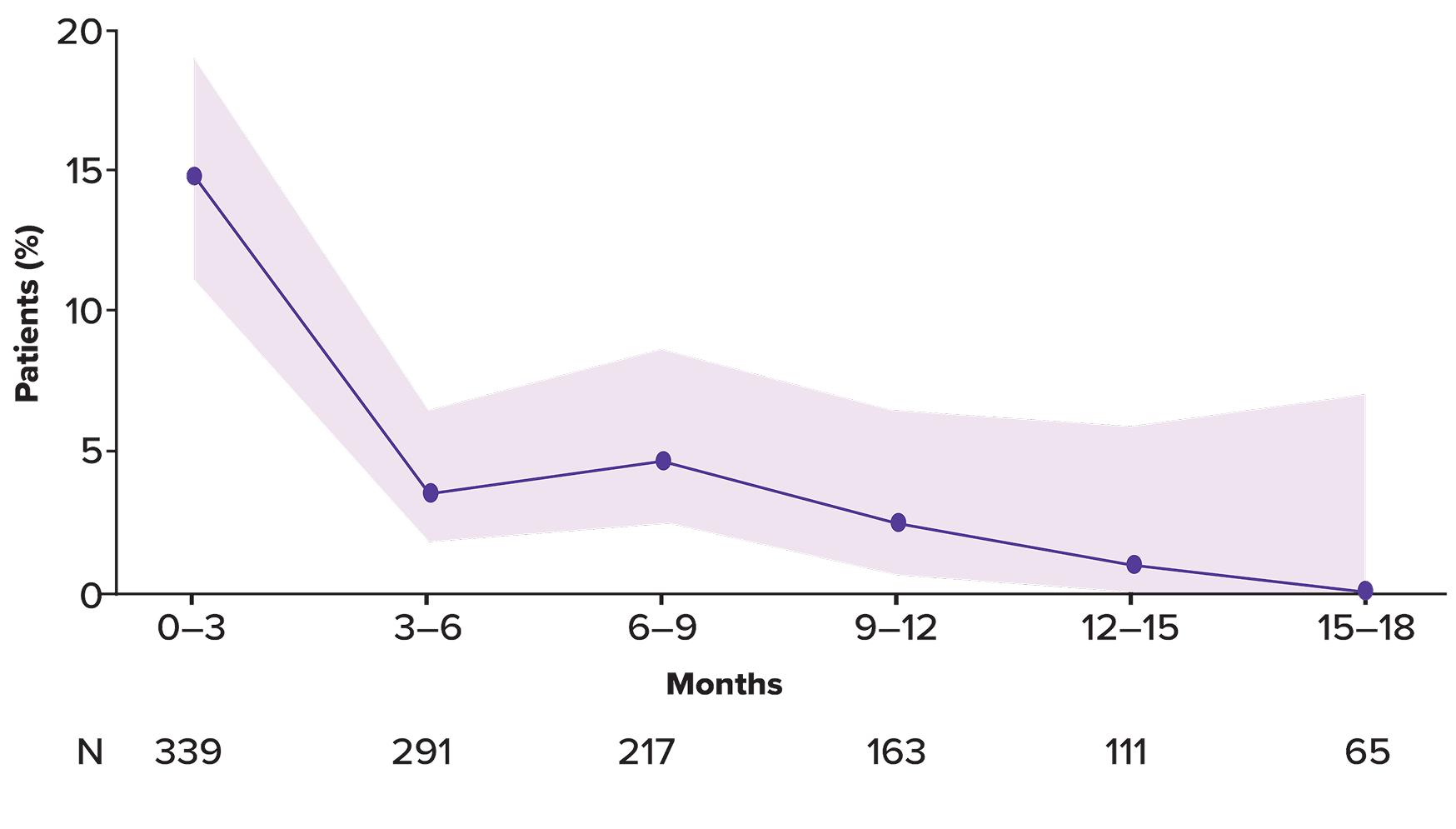
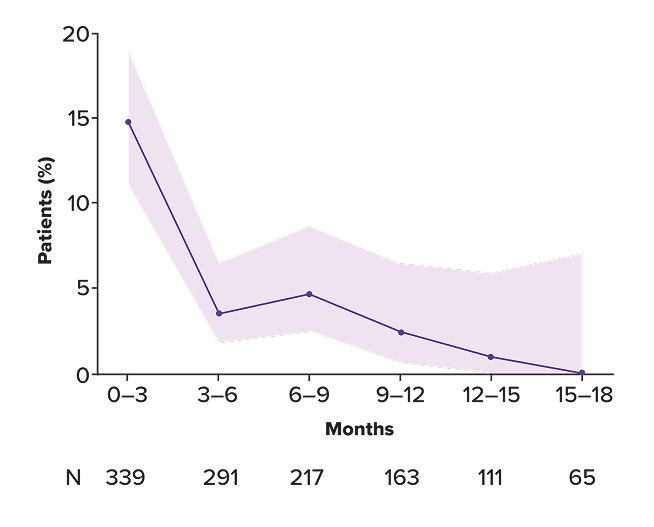
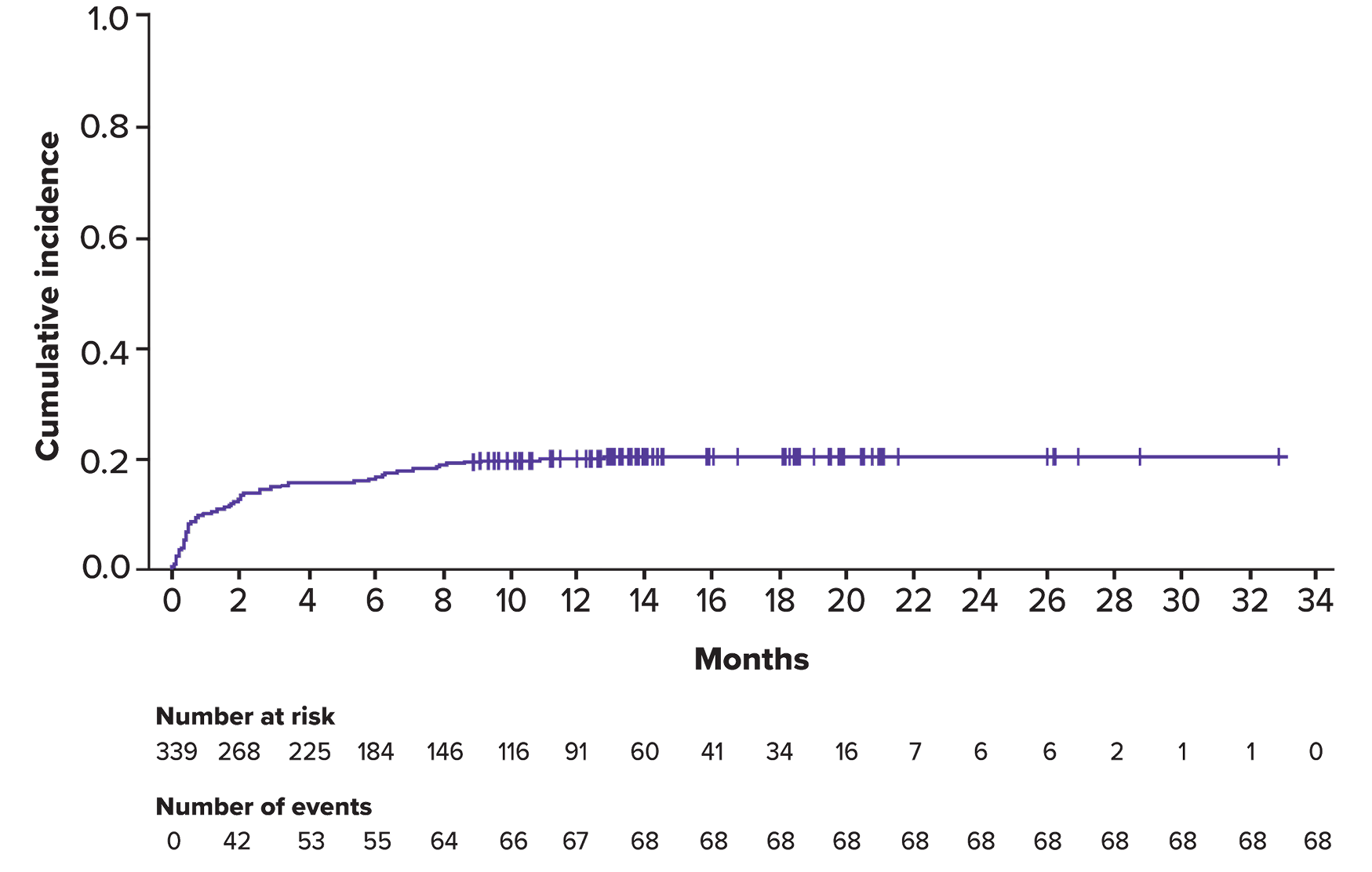

Duration of infections and rates of resolution
Median duration of infections
T-cell redirection–naïve Q2W
12 days (range: 1–201)
n=145
T-cell redirection–naïve QW
11.5 days (range: 2–148)
n=143
T-cell redirection–exposed
12 days (range: 2–51)
n=51
Percentage of infections resolved
T-cell redirection–naïve Q2W
93.8%
n=145
T-cell redirection–naïve QW
94%
n=143
T-cell redirection–exposed
100%
n=51
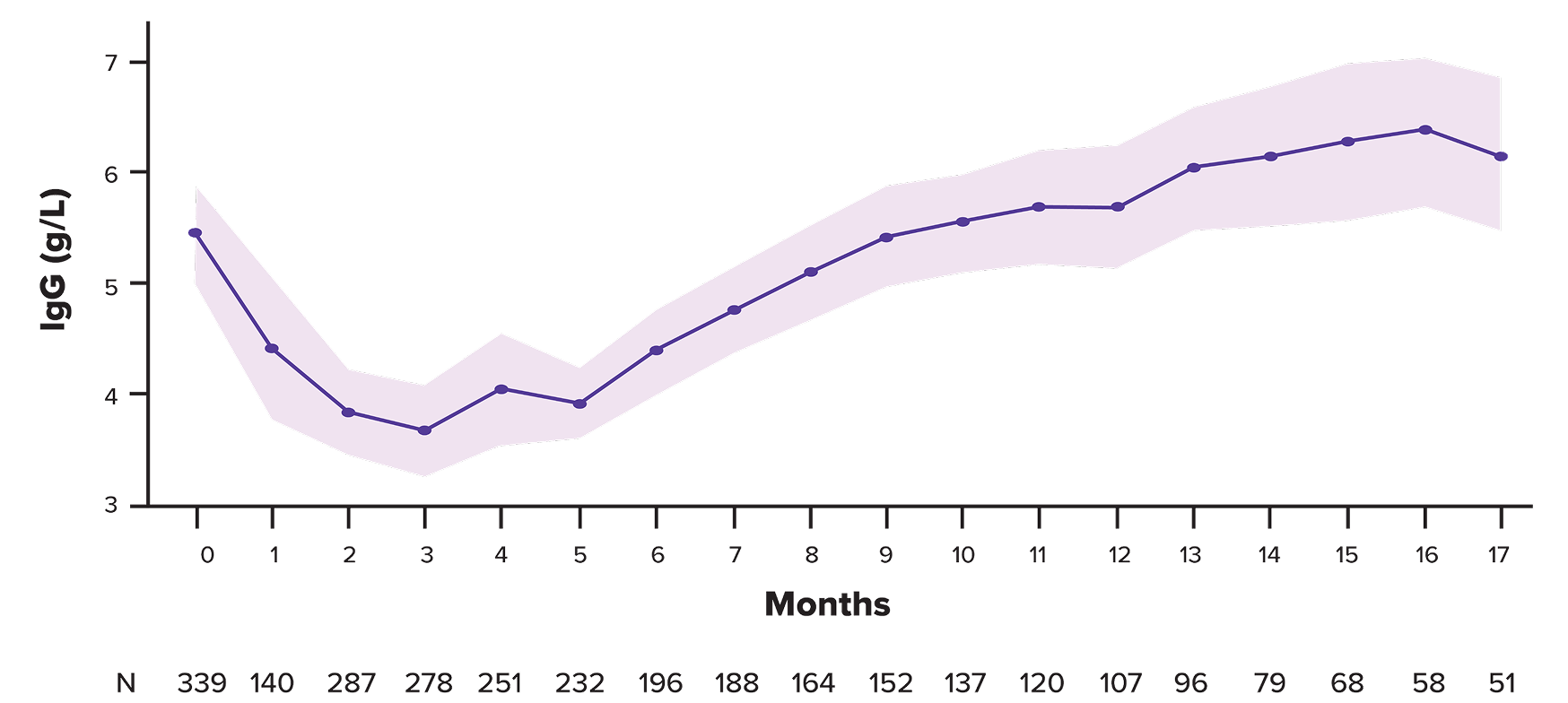
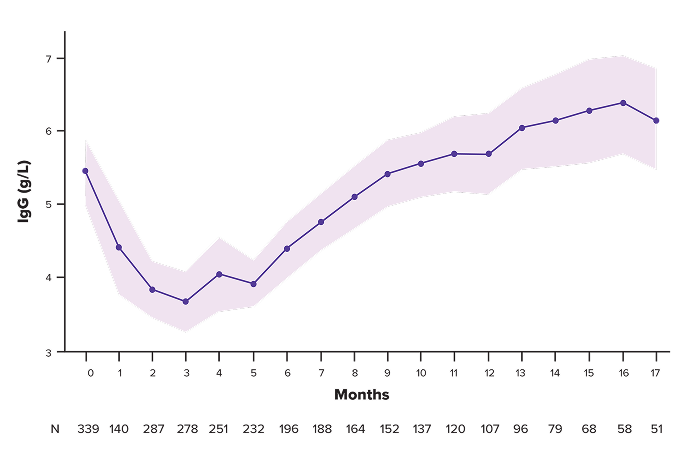
IgG levels recovered after cycle 3 and continued to increase above baseline through cycle 17
Mean polyclonal IgG–adjusted cells during treatment*
IVIG use in MonumenTAL-1
| IVIG use in MonumenTAL-1 | |||
|---|---|---|---|
| Cohort | Total | Received IVIG prior to treatment with TALVEY® | Began receiving IVIG while being treated with TALVEY® |
| T-cell redirection–naïve Q2W | 13.1% (n=145) | 6.2% (n=145) | 6.9% (n=145) |
| T-cell redirection–naïve QW | 14.7% (n=143) | 4.9% (n=143) | 9.8% (n=143) |
| T-cell redirection–exposed | 15.7% (n=51) | 9.8% (n=51) | 5.9% (n=51) |
*lgG was assessed monthly. The majority of samples collected were based on central laboratory testing. Polyclonal lgG was estimated for patients with lgG MM by subtracting M-spike protein values from total lgG values. These calculated values, along with measured lgG levels for patients with non-lgG MM, were assessed to derive mean lgG levels during treatment. Patients who received intravenous immunoglobulin prior to receiving talquetamab were included.
CD, cluster of differentiation; GPRC5D, G protein-coupled receptor class C group 5 member D; IgG, immunoglobulin G; IVIG, intravenous immunoglobulin; MM, multiple myeloma; QW, once weekly; Q2W, every 2 weeks; RP2D, recommended phase 2 dose.
Watch a video detailing infection rates from MonumenTAL-1


See TALVEY®
efficacy data
Learn about
TALVEY® dosing
- Schinke C, Rodriguez-Otero P, van de Donk NWCJ, et al. Infections and parameters of humoral immunity with talquetamab in relapsed/refractory multiple myeloma in MonumenTAL-1. Blood Adv. 2025;9(22):5752–5762. doi:10.1182/bloodadvances.2025016613
- Schinke C, Rodriguez-Otero P, van de Donk NWCJ, et al. Infections and parameters of humoral immunity with talquetamab in relapsed/refractory multiple myeloma in MonumenTAL-1. Blood Adv. 2025;9(22)(Suppl):5752–5762. doi:10.1182/bloodadvances.2025016613