Oral, skin, nail and weight-related adverse reactions seen with TALVEY® in the longer-term follow-up analysis*
Longer-term follow-up of up to 37 months*
You are now viewing a subsequent follow-up analysis of the MonumenTAL-1 trial. This information is not included in the current full Prescribing Information.
LONGER-TERM DATA
| LONGER- TERM DATAAdverse reaction | Adverse reaction | Patients in the MonumenTAL-1 trial | ||
|---|---|---|---|---|
| Total occurrence, n (%) | Leading to dose reduction, n (%) | Leading to discontinuation, n (%) | ||
| Nervous system disorder | ||||
| Dysgeusia† | 273 (72.8) | 21 (5.6) | 21 (5.6)3 (0.8) | |
| Skin and subcutaneous tissue disorders | ||||
| Nail disorder‡ | 215 (57.3) | 3 (0.8) | 0 | |
| Skin disorder§ | 164 (43.7) | 5 (1.3) | 2 (0.5) | |
| Rash∥ | 148 (39.5)¶ | 2 (0.5) | 2 (0.5) | |
| Xerosis | 133 (35.5) | 1 (0.3) | 1 (0.3) | |
| Pruritus | 91 (24.3)# | 2 (0.5) | 0 | |
| Investigations | ||||
| Weight decreased | 152 (40.5)** | 13 (3.5) | 4 (1.1) | |
Note: Dose reduction could mean increasing the interval between doses and/or reducing the overall dose.
*Median follow-up for MonumenTAL-1 cohorts: T-cell redirection–naïve Q2W was >30 months; T-cell redirection–exposed was >28 months; T-cell redirection–naïve QW was >37 months.
†Dysgeusia: ageusia, dysgeusia, hypogeusia, and taste disorder.
‡Nail disorder: koilonychia, nail bed disorder, nail cuticle fissure, nail discoloration, nail disorder, nail dystrophy, nail hypertrophy, nail pitting, nail ridging, nail toxicity, onychoclasis, onycholysis, and onychomadesis.
§Skin disorder: palmar-plantar erythrodysesthesia syndrome, palmoplantar keratoderma, skin discoloration, skin exfoliation, and skin fissures.
∥Rash: dermatitis, dermatitis acneiform, dermatitis contact, dermatitis exfoliative, dermatitis exfoliative generalized, erythema, exfoliative rash, rash, rash erythematous, rash macular, rash maculo-papular, rash popular, rash pruritic, rash pustular, rash vesicular, and stasis dermatitis.
¶Includes 12 (3.2%) Grade 3 or 4 events.
#Includes 1 (0.3%) Grade 3 or 4 event.
**Includes 13 (3.5%) Grade 3 or 4 events.
Patients treated with TALVEY® experienced oral/taste adverse reactions, weight loss, skin-related adverse reactions, and nail-related adverse reactions
- 80% of patients experienced oral toxicities; Grade 3 oral toxicities occurred in 2.1% of patients
- 62% of patients experienced weight loss regardless of having an oral toxicity, with 29% experiencing Grade 2 (≥10%) weight loss and 2.7% experiencing Grade 3 (≥20%) weight loss
- 62% of patients experienced skin reactions; Grade 3 occurred in 0.3% of patients
- 50% of patients experienced nail disorder, with no Grade 3 reactions
Median onset and resolution of select adverse reactions
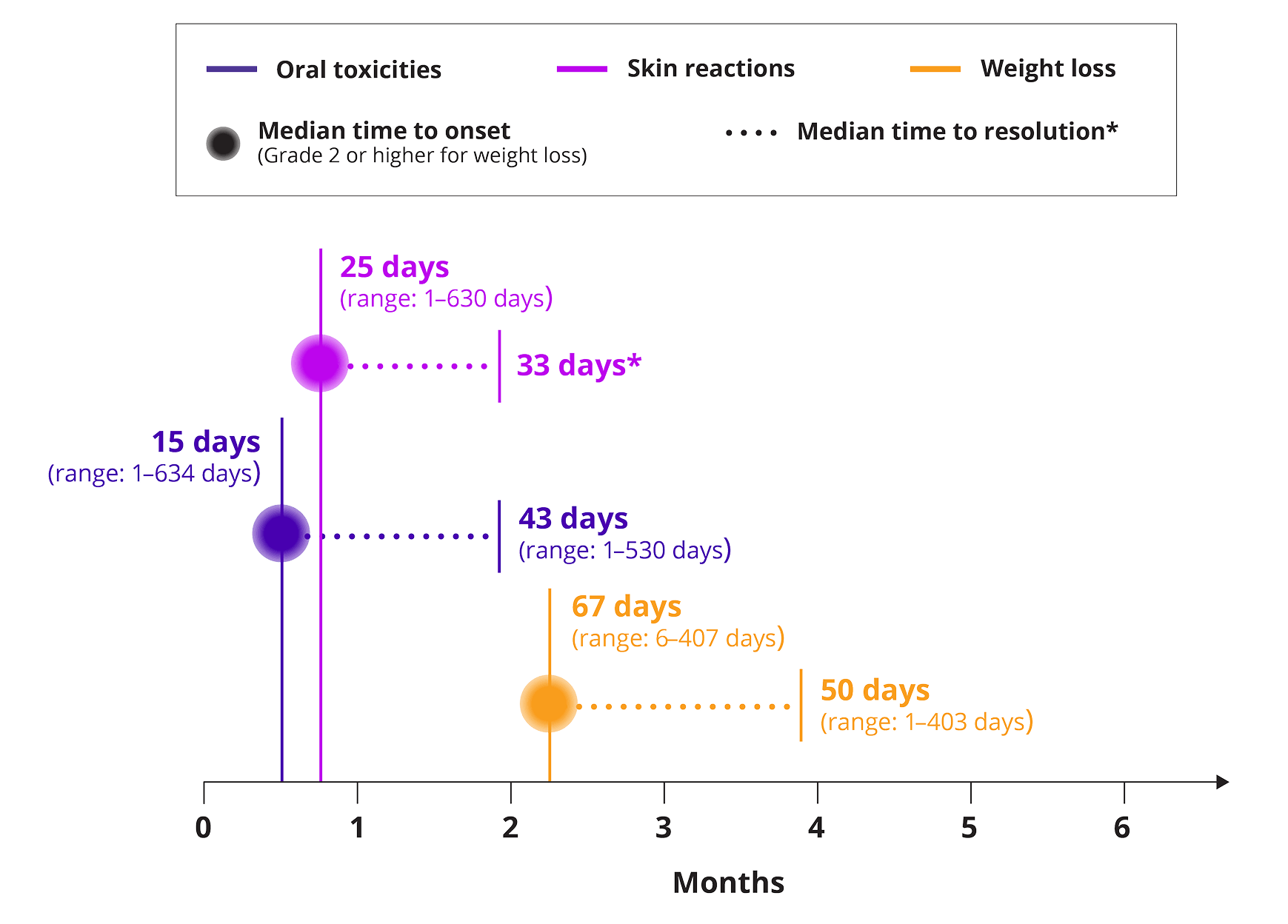
*Resolution of skin reactions was defined as improvement to Grade 1 or less.
- The median time to onset of oral toxicity was 15 days (range: 1 to 634), and the median time to resolution to baseline was 43 days (1 to 530). Oral toxicity did not resolve to baseline in 65% of patients
- The median time to onset of Grade ≥2 weight loss was 67 days (range: 6 to 407), and the median time to resolution was 50 days (range: 1 to 403). Weight loss did not resolve in 57% of patients who reported weight loss
- The median time to onset of skin reactions was 25 days (range: 1 to 630) days, and the median time to resolution* was 33 days
Weight loss over time, associated with BMI
You are now viewing a post hoc analysis of patients with RRMM treated with TALVEY® in MonumenTAL-1. This information is not included in the current full Prescribing Information and should be interpreted with caution.
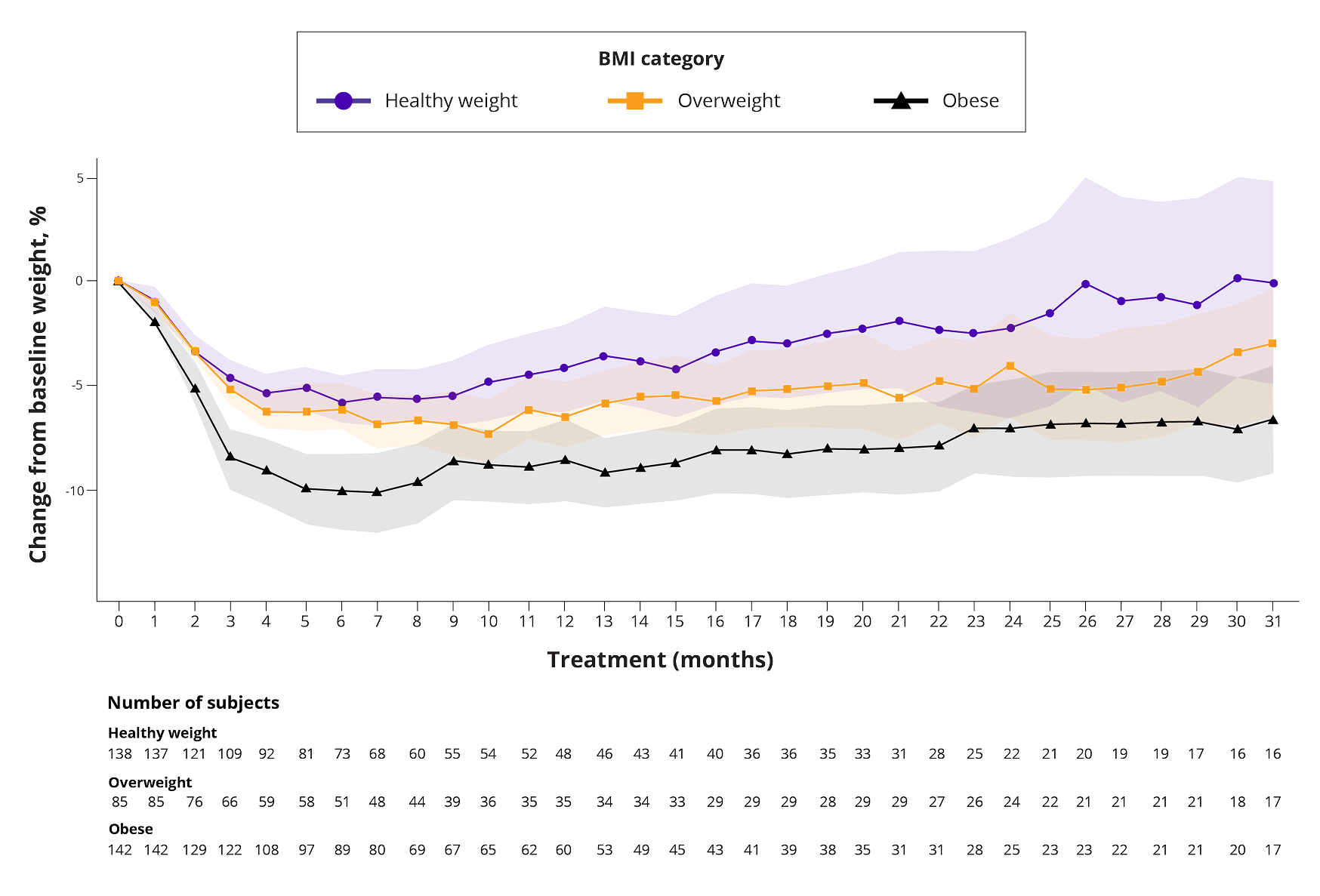
- BMI categories were defined as follows: healthy weight (18.5 to <25), overweight (25 to <30), obese (≥30)
- BMI was calculated as patient body weight (in kilograms) divided by patient height squared (in meters)
- Median follow-up was 30–38 months
Data were plotted as mean percent change from baseline weight. Month 0 represents baseline and month 1 represents the first 30 days of treatment. Shaded areas represent the 95% Cls.
Data plotted if ≥15 patients remained within 30 days of their last TALVEY® treatment and had a measurement within the window. Data were averaged if multiple readings were taken on the same day, and the most recent reading was used if multiple readings were available within the same interval.
Weight loss over time, associated with oral toxicity
You are now viewing a post hoc analysis of patients with RRMM treated with TALVEY® in MonumenTAL-1. This information is not included in the current full Prescribing Information and should be interpreted with caution.
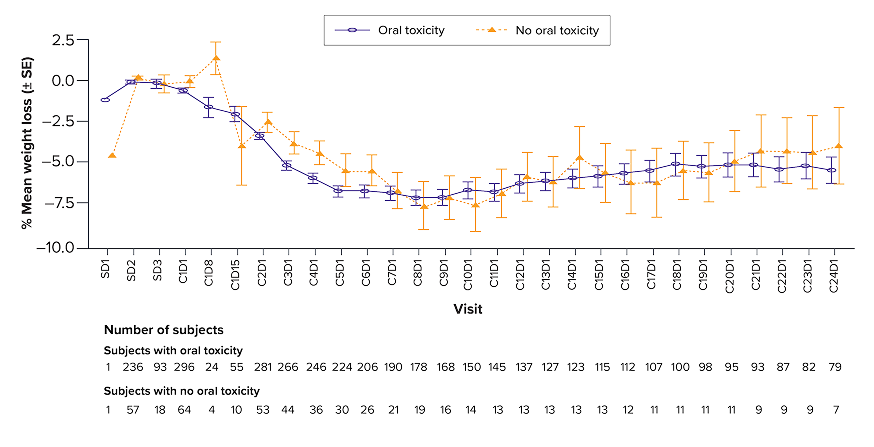
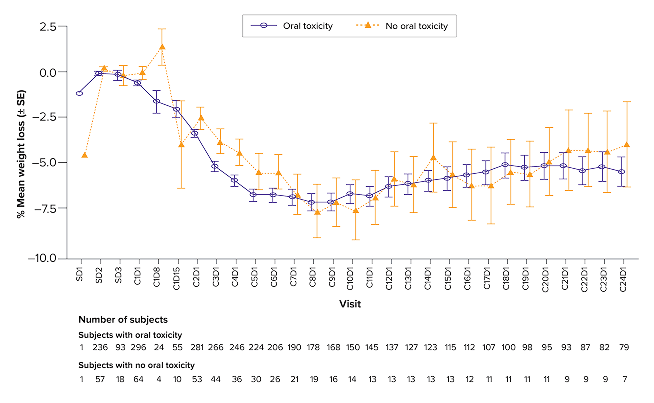
*Includes patients in both the Q2W and QW cohorts. Oral toxicity includes dysgeusia, ageusia, taste disorder, hypogeusia, dry mouth, dysphagia, cheilitis, glossitis, glossodynia, mouth ulceration, oral discomfort, oral mucosal erythema, oral pain, stomatitis, swollen tongue, tongue discomfort, tongue erythema, tongue edema, and tongue ulceration.
Recommendations for managing select adverse reactions
These recommendations were developed by a panel of 37 experts with broad experiences in the management of patients with RRMM. In order to define optimal management strategies, the experts reviewed articles with at least 50 patients enrolled, as well as 2 consensus papers, and held several virtual meetings from 2022 to 2023. Statements with a high agreement (>50%) were incorporated as recommendations.
Oral symptom management: Guidance and recommendations
- Patient education prior to starting therapy about potential on-target, off-tumor adverse events
- Supportive management for oral symptoms
- Dose interruptions or reductions reserved for severe or recurrent cases
- Xerostomia/dysphagia
- Increased hydration (saliva substitutes)
- Sugar-free chewing gum to stimulate saliva flow
- Sodium lauryl sulphate-free toothpaste might be better tolerated
- Dietary modifications may be needed to prevent weight loss
- Nutritional supplements and early nutritional review to optimize oral intake and limit weight loss, especially in patients with a low baseline weight
- Treatment of oral comorbidities (eg, candida, thrush, or nutritional deficiencies leading to glossitis) is encouraged
- Regular dental review to minimize the risk of periodontal disease and caries
Cutaneous AR symptom management: Guidance and recommendations
- Patient education prior to starting therapy about potential on-target, off-tumor adverse events
- Early or prophylactic use of emollients and sunscreen
- Grade 1/2 skin rashes: Low-potency topical corticosteroids (eg, hydrocortisone and triamcinolone), with escalation to medium-potency corticosteroids
- For more extensive (eg, grade ≥3) rashes or rashes refractory to topical therapies
- Short courses of oral steroids (eg, prednisone or prednisolone)
- Longer-term corticosteroids should be avoided where possible due to the risk of infection
- Dermatology consultation for rashes occurring beyond cycle 2 or refractory to emollients or low-potency steroids
Patient counseling information
The recommendations below from The Leukemia & Lymphoma Society and the American Cancer Society may help people living with side effects. This information is not included in the current Prescribing Information and has not been evaluated by the FDA. For more tips, please see lls.org and cancer.org.
Tips for managing mouth problems5
- Maintain good dental hygiene
- Avoid smoking
- Keep mouth moist with hard candy, drinking water, or salivary substitutes
Tips for managing weight loss6
- Maintain a food journal
- Maintain a nutritious diet
- Engage in physical activity
- Your healthcare team may suggest nutrition therapy
Tips for managing skin problems5
- Take warm (not hot) baths or showers
- Pat skin dry
- Use unscented lotion or moisturizing cream
- Wash skin with mild soap and cleansers
- Avoid direct sunlight and apply sunscreen
Tips for managing nail changes5
- Wear gloves when cleaning or gardening
- Avoid biting and picking on nails and cuticles
- Wear comfortable shoes with extra room around the toes
- Keep fingernails and toenails neatly trimmed
- Ask your healthcare provider before you have a manicure
AR, adverse reaction; BMI, body mass index; CI, confidence interval; QW, once weekly; Q2W, every 2 weeks.


- Data on file. Janssen Biotech, Inc.
- TALVEY® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Rasche L, Schinke C, Touzeau C, et al. Efficacy and safety from the phase 1/2 MonumenTAL-1 study of talquetamab, a GPRC5dxCD3 bispecific antibody, in patients with relapsed/refractory multiple myeloma: analyses at an extended median follow-up. Poster presented at: American Society of Clinical Oncology Annual Meeting; May 30–June 3, 2025; Chicago, IL.
- Rodríguez-Otero P, Schinke C, Rasche L, et al. Talquetamab reduced intensity dosing in patients with relapsed/refractory multiple myeloma: MonumenTAL-1 analyses. INCOMPLETE CITATION—NOT YET PUBLISHED
- Side-Effect Management: Caring for Skin, Nails, Hair and Mouth. Leukemia & Lymphoma Society. Accessed October 24, 2025. https://llscorp.dev.lls.org/sites/default/files/National/USA/Pdf/Publications/FF11_SideEffects_Skin_Hair_Nails_Mouth_FINAL.pdf
- Side-Effect Management: Managing Low Appetite and Weight Loss. Leukemia & Lymphoma Society. Accessed October 24, 2025. https://llsorg.widen.net/view/pdf/6obli6epda/en-booklet-side-effects-mgt-appetite-weight-loss-ff15.pdf?t_download=true